Many medical treatments have high initial costs but are considered worthwhile expenses because of the benefits, both direct (improved health and quality of life) and indirect (decreased healthcare spending from avoidance of complications). For treatments such as kidney dialysis, cardiac pacemakers, and organ transplantation, we as a society are willing to pay for treatments with high costs as long as they provide substantial benefits. Cost-effectiveness analysis is a common technique to weigh costs and effectiveness and evaluate whether a treatment that might be expensive has impacts that are large enough to justify the costs, so to speak.
Cost-effectiveness analysis allows comparison of different types of treatments by using a measure of effectiveness called quality-adjusted life-years (QALYs), combining additional years of life (decreased mortality) with increased quality of life (avoiding health complications or motor vehicle crashes, for example). The cost per QALY measures the cost that is associated with each additional year of life with perfect health. This is not the same as adding another year of life, as it incorporates not only the additional years of life and the quality of those additional years. For example, if a treatment allows someone to live 5 years longer but at a very poor quality of life (0.10 on the scale), the improvement would be less than 1 QALY (5 years x 0.10 = 0.5 QALY). For cost-effectiveness, the cost of the treatment is then divided by the change in QALYs to give the cost per 1 QALY gained.
Sleep Apnea Treatment and Cost Effectiveness
We are fortunate that continuous positive airway pressure (CPAP) is relatively inexpensive, as it can be extremely beneficial for patients who wear it and sleep comfortably with it. However, we know that many patients do not tolerate or benefit from CPAP. These patients should consider alternative treatments, including surgery or oral appliances. These are generally covered by medical insurance in the United States because they are supported by research studies demonstrating benefits that balance the costs.
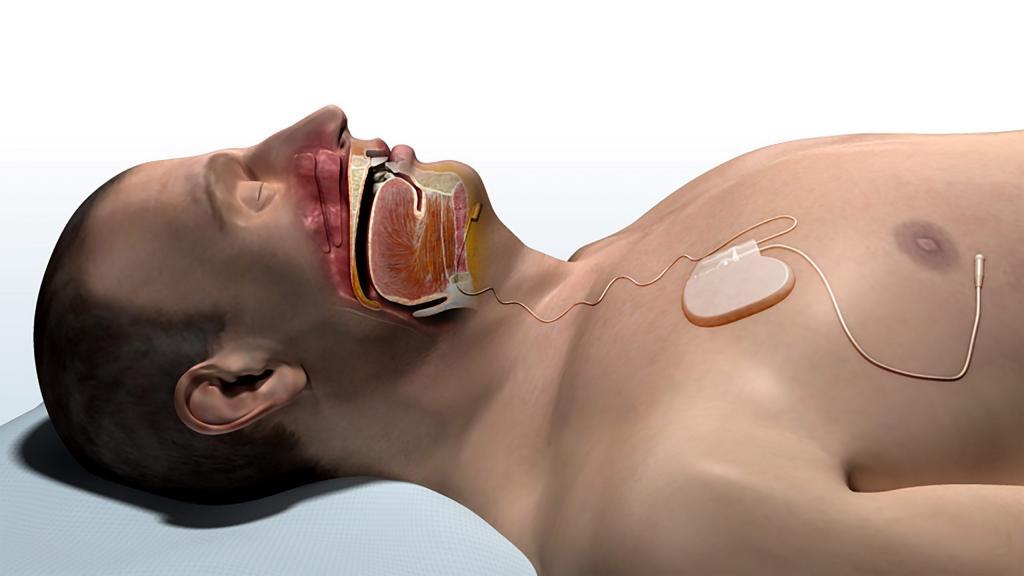
Inspire Medical’s Upper Airway Stimulation therapy has initial costs that are higher than most other treatments for obstructive sleep apnea. As happens with many new treatments—especially those with high initial costs—it is important to compare costs and effectiveness to see how it compares to other well-accepted treatments. The May 2015 issue of the medical journal SLEEP includes a cost-effectiveness analysis of Upper Airway Stimulation on which I am an author.
How was the study of Upper Airway Stimulation costs done?
This study was designed to provide a general estimate of the costs and effectiveness of Upper Airway Stimulation. We used established cost-effectiveness analysis methods. Direct healthcare costs (costs paid by medical insurance) were almost $43,000; these were the Medicare payment amounts for healthcare costs like the surgical implantation procedure and additional sleep studies and office visits, minus the cost savings for reducing potential sleep apnea complications (heart attack, stroke, and motor vehicle crashes). To be conservative, we did not include financial benefits that may be seen with sleep apnea treatment, such as improved worker productivity, that can occur with sleep apnea treatment.
The effectiveness evaluation was based on the STAR Trial publication, in which 66% of patients receiving the device achieved a dramatic improvement or resolution of their sleep apnea. The trial data were used to estimate the reduction in adverse outcomes (mortality, heart attack, stroke, and motor vehicle crashes), based on published studies of other sleep apnea treatments. These outcomes were combined to show that Upper Airway Stimulation provides 1.09 QALYs over a person’s lifetime.
What is the cost effectiveness of Upper Airway Stimulation?
Combining costs and effectiveness, the study showed that the lifetime incremental cost-effectiveness ratio for Upper Airway Stimulation was about $40,000 per QALY. Cost-effectiveness analyses are never entirely precise, so we used two common approaches to account for uncertainty in certain parts of the model: sensitivity analysis and Markov estimation. Again, to be conservative, we used a wide range (±30%) in the estimates for the cost components and a wider range for the effectiveness estimates. Incorporating the variation did not markedly change the conclusions; 70% of the cost-effectiveness ratio estimates were below $50,000 per QALY, and 95% of estimates were below $100,000 per QALY. Therefore, the results show that Upper Airway Stimulation has favorable cost-effectiveness, as the generally-accepted threshold for medical treatments has been considered to be $50,000-100,000 per QALY, although some have argued that $100,000-150,000 is more appropriate.
This study has limitations, like any research. Dr. Edward Weaver from the University of Washington and Dr. Jonathan Skirko from the University of Colorado wrote an editorial in the same issue, pointing out some of these. I have known Ed for many years as a mentor, colleague, and friend, and I have always welcomed (and continue to welcome) challenging opinions. In fact, many of their concerns were included in our paper. They felt our study translated data on changes in the apnea-hypopnea index in a certain group of patients to health outcomes (such as heart attack and stroke) that were not measured directly. We certainly made best efforts and used published studies that make these same translations for other sleep apnea treatments. Our approach is common to those adopted by similar studies of other medical devices in all areas of medicine. In my opinion, the most important limitation regarding this study is that the effectiveness data come from a single study and use data from 12 months to project results over 10 years. While a perfect world would include multiple studies tracking thousands of patients over decades, we will likely never have these studies. Instead, we accounted for uncertainty in multiple ways and relied on previous research showing that the benefits of Upper Airway Stimulation are maintained at 18 months for those who respond initially. Data from 2 years after Upper Airway Stimulation system implantation will be presented at the upcoming SLEEP meeting in June 2015, and I look forward to seeing them.
The bottom line
Nobody should think that Upper Airway Stimulation has a precise cost-effectiveness ratio of $40,000 per QALY. Instead, this study showed that Upper Airway Stimulation has a cost-effectiveness ratio that is well within the range used to cover many medical interventions, even those with high initial costs. For appropriate patients, this is an exciting treatment option with favorable cost-effectiveness.
If you are interested in learning whether Inspire Medical’s Upper Airway Stimulation therapy is right for you, please do not hesitate to contact me at [email protected] to schedule a video consultation or to ask any questions.
Disclosure: I treat patients with obstructive sleep apnea with the Upper Airway Stimulation system. However, I do not own any part of the company and do not receive any royalties from sales of their devices. I have been paid for my work assisting Inspire’s preparation for their 2014 presentation to the United States Food and Drug Administration and the development of the cost-effectiveness model discussed above (but not the performance of the cost-effectiveness analysis or writing of the paper).




Clark Jackson says:
What is the approximate initial cost? CPAP works for me but sometimes does not for 3-4 days at a time. I might consider in investing in Inspire therapy
Dr. Kezirian says:
Inspire Upper Airway Stimulation is fairly expensive, but the key is that we would work with (and, if necessary, against) your insurance company to do everything possible to get it approved and covered. If you are not doing well with CPAP (I cannot entirely tell, based on what you have written), then we should at least consider other treatment options, including Inspire Upper Airway Stimulation. If you would like to discuss this in more detail, please feel free to contact me.
ALLAN R BURMAN says:
What is the cost for the Upper Airway? I’ve heard the cost can run $40,000. Does Medicare cover the cost?
Dr. Kezirian says:
The cost without insurance is about $45,000 at our medical center, which is about half of what I have seen as estimates from some other centers. As far as Medicare, it is all still up in the air. They have not issued a clear statement (called a Coverage Determination), but apparently they are considering what to do. For now, Medicare coverage is on a case-by-case basis. The difficulty is that you only learn of coverage after having the procedure (and likely appealing an initial denial) because Medicare does not provide preauthorization like many private insurance programs.
Christopher Reed says:
I am a 100% disabled veteran from the VA I use a CPAP will my VA insurance for my Tricare Prime insurance cover this procedure?
Dr. Kezirian says:
It may, but you would want to check with your VA sleep physician to see if they are an Inspire center or know of one close to you.
Andrea Talley says:
In lieu of having Orthognathic surgery, this sounds like it might help without so much downtime. I’d like more info or reviews over a longer period than 18 months.
Does this mean a 22 year old candidate would have to wear this for the rest of their lives, changing the battery every 11 years?
Dr. Kezirian says:
The main unit (called the pulse generator) has a battery that lasts about 11 years. This portion of the system would need to be replaced, but the two other component would not need replacement.
Studies of the Inspire Upper Airway Stimulation sytem have been conducted at 5 years after implant, showing consistent benefits (results stable at 1, 2, 3, 4, and 5 years) for most patients.
Andrea Talley says:
Dr. Kezirian,
Where could we get this done? Are there physicians here in Greenville, SC?
We have Cigna insurance? Do you accept Cigna?
What are some of the more common side effects besides a sore throat for a few moments when waking up? Any other that affect more people than just one or two?
What kind of complications can happen? Severity of those complications long term?
Dr. Kezirian says:
We do accept CIGNA, but there also are surgeons who have undergone Inspire training closer to you. You can find them on the Inspire website: inspiresleep.com. I would be happy to see you, so please e-mail me if you would like to schedule an appointment.
Kylie Brown says:
How much is the cost of the new Nyxoah product as im very interested please Thanku
Dr. Kezirian says:
It is only available through trials, so there is no price at this time.
DENISE says:
Everything I read is pretty vague….does insurance ever agree to coverage?
Dr. Kezirian says:
Absolutely. It is not automatic, however.
James Lloyd says:
Have you had any luck working with Kaiser to cover the implant?
Dr. Kezirian says:
To my knowledge, Kaiser does not cover the Inspire therapy for sleep apnea. I have had some patients switch away from Kaiser for sleep apnea surgery (to see me and have it covered). The Inspire therapy is certainly not always covered by insurance outside the Kaiser system, but at least it is an option. Also, I am not aware of any Kaiser surgeon in southern California who performs the full spectrum of procedures for sleep apnea.
Neil M says:
How about United Health Care?
Dr. Kezirian says:
United Health Care has definitely covered Inspire Upper Airway Stimulation. Generally, insurance company approvals have involved an initial denial, followed by an appeal within the insurance company that is denied, followed by an appeal for an external review (sometimes called an Independent Medical Review) where the procedure is often approved if the patient is an appropriate candidate. Aetna is the only insurer that has a policy providing coverage (assuming a patient meets the qualifying criteria).
Ruth Lewis says:
I would like to hear more about this implant. I’m 71 year old overweight with sleep apnea, COPD, kidney disease, insulin dependent diabetic, non alcoholic cirrhosis liver, enlarged spleen, arthritis of spine and joints, neuropathy of feet, legs and hands, Chronic heart failure. I would like to be able to sleep all night. I have UPMC health insurance in Uniontown, Pa
Dr. Kezirian says:
You can go to the company’s website at inspiresleep.com for more information.
Doug Tidwell says:
Does Medicare cover all the costs of getting this?
Dr. Kezirian says:
Medicare currently does not have a clear policy (called a Coverage Determination) on this treatment, although they do cover most treatments for sleep apnea.
Nancy Cooper says:
Any knowledge of Tri care prime covering this procedure?
Dr. Kezirian says:
TRICARE has definitely covered this, but approvals now are still on an individual basis. The first step is to proceed with an evaluation to see if it is the right approach for you. Then we would submit to TRICARE for approval. They have definitely covered this for many patients, but we would first need to show clearly that you are the right kind of patient who would benefit from it.
Kelly says:
Ok I have Tricare Prime. Does prime cover this device? I am interested in finding out.
Dr. Kezirian says:
Coverage has happened for many insurance plans around the country. Sometimes it is covered under their plan policies, but sometimes there is a need to go through an authorization process. I am not sure of the exact policy for Tricare Prime, but you should start by seeing someone at an Inspire center if you are interested. I would be happy to see you myself.
Donnie says:
I have blue cross ins. Will it cover the cost of the implant.
Dr. Kezirian says:
Approval for Upper Airway Stimulation still occurs on a case-by-case basis, meaning that the first step is to proceed with an evaluation to see if it is the right approach for you. Then we would submit to your insurance company for approval. Blue Cross definitely has covered this for many patients, but we would need to show clearly that you are the right kind of patient who would benefit from it.
sharon arpano says:
i have medicare & harvard pilgrim ins.. covered??
Dr. Kezirian says:
Right now, Medicare coverage is up in the air. You will want to find a surgeon or center offering this and speak with them.
Mike Crain says:
Does Medicare cover all expenses or will I incur any out-of-pocket expenses?
Dr. Kezirian says:
Right now, it is not clear that Medicare covers Upper Airway Stimulation. That may change, as I am told they are going through a major effort to make a national coverage determination. Right now, Aetna is the only insurer that is clearly covering Upper Airway Stimulation for patients that meet key criteria where this technology has shown clear benefits.
Laura Felde says:
Great info, thanks Dr. Kezirian! Do you know if United Health Care has covered this treatment?
Dr. Kezirian says:
They have, although it can still require appeals of initial denials and is not guaranteed.
Bryan Keck says:
Does Medicaid cover the Inspire device?
Dr. Kezirian says:
Not to my knowledge.
Carly Simone says:
What about Aetna Elite PPO (Medicare PPO)?
Dr. Kezirian says:
If a Medicare plan provides preauthorization for procedures, a surgeon can submit for preauthorization and find out if there will be coverage. The challenge in dealing with traditional Medicare is that they do not provide preauthorization information, so you can be left not knowing whether they would cover it after the surgery (and then be responsible to pay for it yourself).
Tom says:
What about Medicaid? Will it cover it?
Dr. Kezirian says:
Not at this time, to my knowledge.
Christopher B Lane says:
Will Blue Cross Blue Shield HDHP cover it. My insurance plan goes as follows: Anything that’s preventable usually they will cover it. In Network covers 90/10. I was curious if will cover the treatment option. I am currently on the CPAP machine for over 3 years now.
Dr. Kezirian says:
If you are using CPAP and doing well with it, you should stay with it. If you are not, you should see a team that provides Upper Airway Stimulation to determine if you would be a good candidate. You would then go through the process of obtaining insurance authorization. Most insurance plans do cover it, although it can require initial denials that are followed by appeals to get authorization.
Kathleen A McDermott says:
Was I talking to you on Facebook last year about Medicare not covering the inspire therapy after my endoscopy procedure in Syracuse, New York? I want you to know I took your advice and found a doctor in my home town in Rochester, NY. The doctor’s name is Sveta Karelsky, MD and after my endoscopy procedure at UofR Medicine at Highland Hospital December 19, 2018, I will get the results January 14, 2019 and request coverage from Medicare AGAIN. It may be approved now that many more have been done.
Dr. Karelsky told me she has done five (5) so far. Is that enough experience? I guess the next question would be is, “Were they successful?” I wonder what Medicare’s acceptable number is because they said it was too experimental. The Syracuse doctor retired, but I think he did ONE OTHER PERSON. So Medicare may have done me a favor by saying no, initially.
Who can I talk to who has had it done?
I look forward to your reply, Dr. Kezirian.
Dr. Kezirian says:
Sveta Karelsky actually came out and spent some time with me a few years ago when I was at UC San Francisco. Inspire was not available at that time, but she was great. I would expect that 5 cases should be sufficient experience for someone like her. The issue is the uncertainty of Medicare coverage at this point in time.
Rachel Cooley says:
I am interested if you have ever had someone with MVP medicare advantage Plan, insurance covered this procedure? Also, I suffer from Insomnia, would this help or maybe hinder the insomnia? What is your experience.
Dr. Kezirian says:
Medicare does not issue preauthorization, and the coverage for Medicare patients has become more straightforward. Insomnia can be a major challenge with Upper Airway Stimulation if it is substantial and not well-controlled. Insomnia can make it difficult for a patient to become comfortable with the stimulation.
Charlie Montante says:
Does independent health cover this
Dr. Kezirian says:
Aetna is the only payor that covers Upper Airway Stimulation in patients meeting the core criteria without needing appeals. Many other payors issue an initial denial, but the chances of coverage after going through an appeals process have been improving over the past 1-2 years.
Michele says:
I just came from my doctor’s office, and he told me that cencal ( medi-cal ), would cover my procedure. Is that true?
Dr. Kezirian says:
I am not aware of Medi-Cal (or Medicaid) covering this.
Steven cohen says:
Is this device approved in Canada specifically Ontario?
Dr. Kezirian says:
It is not approved in Canada, to my knowledge.
Elizabeth says:
Im active duty military. Have you had any luck getting approval for those of us in the military? I’ve had sleep apnea for over ten years and two CPAP machines and dozens of masks I just can’t get used to it. I’ve given up hope of getting a good nights sleep and resolved in my mind that is probably how I’ll die.
Dr. Kezirian says:
Absolutely you would want to be treated for your sleep apnea. For the right candidates, many payors have approved Upper Airway Stimulation. I had have approvals through the VA but not for active duty military, but I have had many types of procedures approved for active duty military patients and cannot imagine that this would be so different.
Harvey kokemor says:
What are the complications and risks in this elective surgical procedure? What are the healing times and limitations after surgery? Is there any prescription drugs needed with the surgery ?
Carol says:
I am also interested in the risks of the inspire surgery.
Dr. Kezirian says:
You can learn more about them on my website page that discusses the procedure: https://sleep-doctor.com/surgical-treatment-overview/upper-airway-stimulation/.
Audrey Nebel says:
Typically how many appointments are required for the initial testing and what is the required follow treatment for the Inspire surgery? I do not have a facility close that does this surgery so that is a consideration for me
Dr. Kezirian says:
When patients travel to see me, there is an initial office visit, drug-induced sleep endoscopy procedure in the operating room, and then the surgery, with an overnight stay in the hospital. There are usually several follow up visits and tests with the sleep medicine team, so in some cases I will see patients to implant the device and then look for a center closer to their home for the sleep medicine visits and tests.
Patti Hansen says:
Does Humana cover this therapy?
Dr. Kezirian says:
A number of major insurance companies cover this directly if you meet standard criteria. Humana is not one of those with automatic coverage, but this just requires a submission for preauthorization, with appeals if the request is denied. I forget the exact number, but well over 100 insurance companies have covered Inspire therapy for patients, even if it has required this preauthorization/possible appeal process.
Stella Lowe Mcdonald RN CCM says:
I am a retired RN in my mid 60s with known severe osa. HX of mi with beginnings of some diastolic heart failure have tried so many masks, pillows and pads without success. I just can’t sleep. It’s almost like my nose has a horrible itching reaction to all of them. I formerly was an insurance co case Mgr and-am familiar with much of the procedures and criteria that must be met. I know we have a physician at Vanderbilt that you list. I have Blue Cross medicare extra diamond. Have you had any interaction with vanderbilts program andB.C TN? Thank yoy
Dr. Kezirian says:
One of our former fellows is a sleep surgeon at Vanderbilt: Dr. David Kent. He is excellent.
Lana says:
Can you qualify if you have sleep apnea as well as narcolepsy?
Dr. Kezirian says:
It is possible but not the most common situation.
Rob says:
I have Blue Advantage HMO. Have you had any success with them? Also, does a patient have any restrictions on awake activities such as intense exercise, weightlifting, etc?
Dr. Kezirian says:
There has been success with almost all payors. There should be no restrictions on intense exercise and weightlifting or almost all other activities.
Mr Willis says:
Thank you for providing this information. I was very excited about Inspire, after struggling with CPAP (three times) and oral appliances that fail to treat severe (63 obstructions/hr ?) OSA that I’ve had since I was a teenager.
I was told by my Doctor (who is certified for Inspire procedures, but has never actually performed this surgery on a living human yet) that I qualified, but based that on Medicare BMI guidelines. Aetna rejected this, however, as they use a different BMI limit. Net-net – Aetna won’t cover the surgery until I lose 30-40 lbs. I am not morbidly obese – I wear size 36 waist, but should wear about a 34. I am certainly thinner than several of the people who gave testimonials on the Inspire website.
I am extremely frustrated… the initial test (when they put you under) came with a very high out-of-pocket cost, and now that’s all for naught.
My advice: before spending a single cent, follow up with your insurance company AND Inspire DIRECTLY. Don’t get mislead my a doctor who is not the final authority on your approvals.